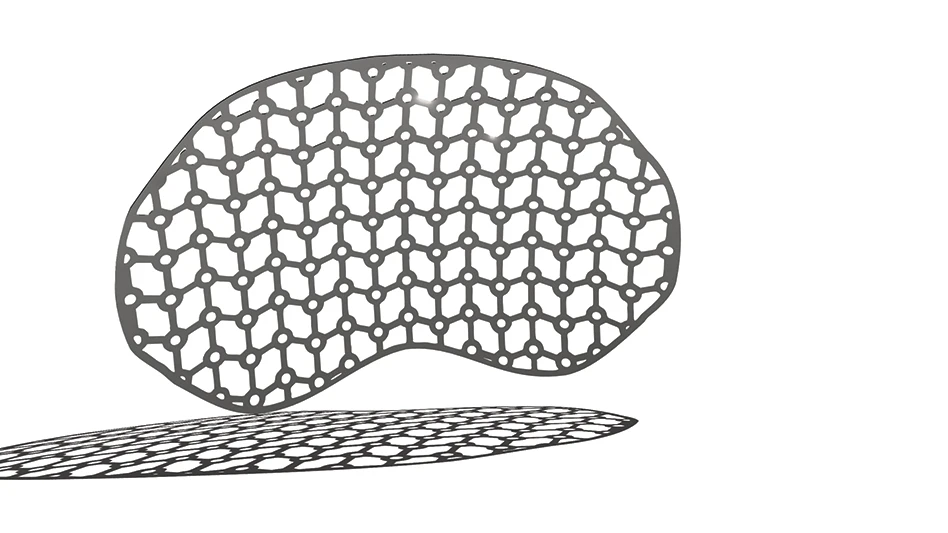
Cagent Vascular received FDA 510(k) clearance of its Serranator PTA Serration balloon catheter for treating below-the-knee (BTK) lesions. The Serranator angioplasty balloon device is FDA cleared and CE marked, embedding serration technology into a semi-compliant balloon for treating peripheral artery disease (PAD). It’s designed to create multiple longitudinal lines of interrupted micro-serrations to aid arterial expansion.


Explore the July 2020 Issue
Check out more from this issue and find your next story to read.
Latest from Today's Medical Developments
- Stryker’s flexible syndesmotic fixation device stabilizes ankle injuries
- Mergers & acquisitions news: MGS, Quantum Surgical bolster medtech portfolios
- Exchangeable-head solid carbide cutting tools
- NextDent 300 MultiJet printer delivers a “Coming of Age for Digital Dentistry” at Evolution Dental Solutions
- Get recognized for bringing manufacturing back to North America
- Adaptive Coolant Flow improves energy efficiency
- VOLTAS opens coworking space for medical device manufacturers
- MEMS accelerometer for medical implants, wearables