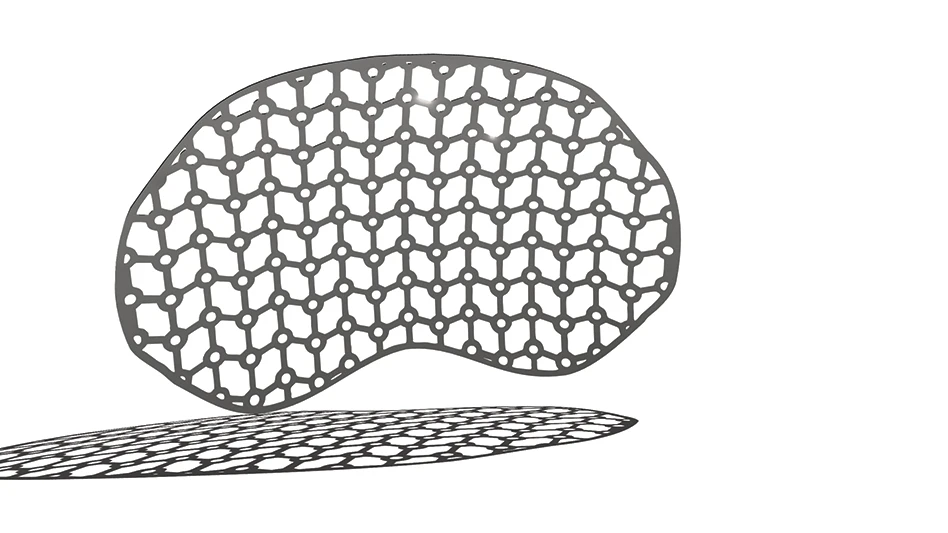
Researchers at Stony Brook University, Stony Brook, NY, have developed a device that can be used to optimise cardiovascular device design to eliminate the risk of clot formation.
The device thrombogenicity emulator (DTE) measures the potential that a cardiovascular device will cause a blood clot.
During the Phase I of the project, the researchers developed and tested the device in laboratory models. In the Phase II study, the researchers are planning to use DTE to identify the areas in cardiovascular devices where clots can form.
Lead researcher Danny Bluestein said that within the DTE, it is possible to tweak the geometry of the device's design to optimise it and minimise or eliminate these hot spots.
Latest from Today's Medical Developments
- Stryker’s flexible syndesmotic fixation device stabilizes ankle injuries
- Mergers & acquisitions news: MGS, Quantum Surgical bolster medtech portfolios
- Exchangeable-head solid carbide cutting tools
- NextDent 300 MultiJet printer delivers a “Coming of Age for Digital Dentistry” at Evolution Dental Solutions
- Get recognized for bringing manufacturing back to North America
- Adaptive Coolant Flow improves energy efficiency
- VOLTAS opens coworking space for medical device manufacturers
- MEMS accelerometer for medical implants, wearables