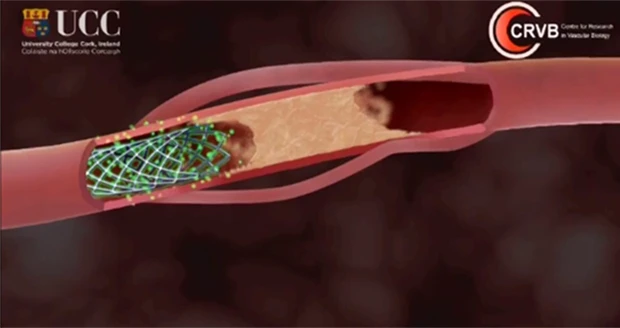
Merit Medical Systems Inc., a leading manufacturer and marketer of proprietary disposable devices used primarily in cardiology, radiology, and endoscopy, received 510(k) clearance from the Food and Drug Administration to market the Merit Laureate hydrophilic guide wire.
In February 2012, Merit Medical received a warning letter from the FDA regarding modifications in the manufacturing process for which the FDA required additional information. Merit complied by filing a new 510(k) submission.
Merit discontinued sales of the Merit Laureate during the review period in the United States, but continued to market the product, which is manufactured in Galway, Ireland, in international markets.
"We are pleased to conclude this process and provide this product immediately to our U.S. customers," states Fred P. Lampropoulos, Merit Medical's chairman and CEO. "We believe this segment of the guide wire business offers substantial opportunity and intend to introduce additional hydrophilic wires upon regulatory clearance in the near future."
Latest from Today's Medical Developments
- NextDent 300 MultiJet printer delivers a “Coming of Age for Digital Dentistry” at Evolution Dental Solutions
- Get recognized for bringing manufacturing back to North America
- Adaptive Coolant Flow improves energy efficiency
- VOLTAS opens coworking space for medical device manufacturers
- MEMS accelerometer for medical implants, wearables
- The compact, complex capabilities of photochemical etching
- Moticont introduces compact, linear voice coil motor
- Manufacturing technology orders reach record high in December 2025





