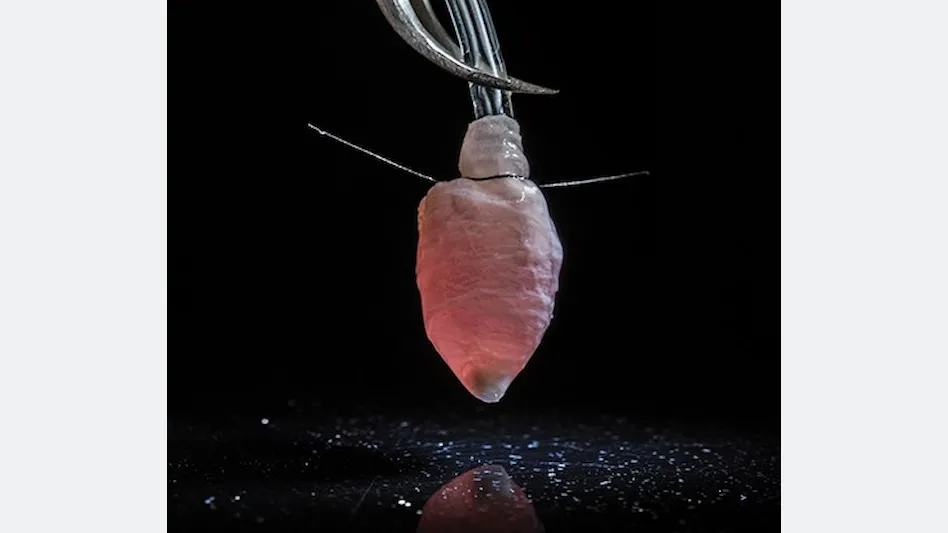

A three-dimensional model of a human heart left ventricle will allow researchers to study heart function with many of the same tools used in the clinic, including pressure-volume loops and ultrasound, and the 3D model could be used to study diseases, test drugs, and develop patient-specific treatments for heart conditions. Bio-engineered by Harvard University researchers, the tissue was a nanofiber scaffold seeded with human heart cells, and the scaffold acts like a 3D template, guiding the cells and their assembly into ventricle chambers that beat in vitro.
“Our group has spent a decade-plus working up to the goal of building a whole heart, and this is an important step toward that goal,” says Kit Parker, the Tarr Family Professor of Bioengineering and Applied Physics at the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS), senior author of the study.
“The long-term objective of this project is to replace or supplement animal models with human models and especially patient-specific human models,” says Luke MacQueen, first author of the study and postdoctoral fellow at SEAS and Harvard’s Wyss Institute of Biologically Inspired Engineering.
The key to building a functional ventricle is recreating the tissue’s unique structure. In native hearts, parallel myocardial fibers act as a scaffold, guiding brick-shaped heart cells to align and assemble end-to-end, forming a hollow, cone-shaped structure. When the heart beats, the cells expand and contract like an accordion.
To recreate that scaffold, the researchers used a nanofiber production platform known as pull spinning, developed in Kit Parker’s Disease Biophysics Group. A high-speed rotating bristle dips into a polymer reservoir and pulls a droplet from the solution into a jet. The fiber travels in a spiral trajectory and solidifies before detaching from the bristle and moving toward a collector.
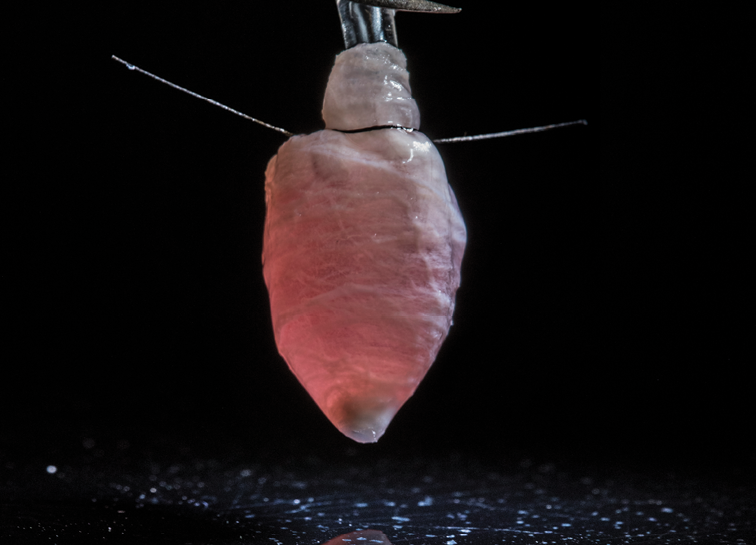
To make the ventricle, the researchers used a combination of biodegradable polyester and gelatin fibers that were collected on a rotating collector shaped like a bullet. Because the collector is spinning, all the fibers align in the same direction.
After building the scaffold, the researchers cultured the ventricle with either rat myocytes or human cardiomyocytes from induced stem cells. In three-to-five days, a thin wall of tissue covered the scaffold and cells were beating in sync. From there, researchers could control and monitor the calcium propagation and insert a catheter to study the pressure and volume of the beating ventricle.
Using human cardiomyocytes from induced stem cells, the researchers were able to culture the ventricles for six months and measure stable pressure-volume loops.
Next, the researchers aim to use patient-derived, pre-differentiated stem cells to seed the ventricles, which would allow for more high-throughput production of the tissue.
Harvard University, John A. Paulson School of Engineering and Applied Sciences (SEAS)
https://www.seas.harvard.edu

Explore the November December 2018 Issue
Check out more from this issue and find you next story to read.
Latest from Today's Medical Developments
- Tsugami America’s Technical Center in Minnesota
- RobOps Copilot for AI-powered robot optimization
- US companies invest heavily in robots
- #34 Lunch + Learn Podcast - Cobots' potential to revolutionize aerospace manufacturing with Techman Robots
- Universal Robots announces seamless integration with Siemens PLCs
- This month's Manufacturing Lunch + Learn is May 16
- Ultrahuman wearable technology
- Additive Manufacturing for Aircraft Cockpit Interior Components